What is the meaning of Water Hardness?
Water Hardness: Hard water is water that contains a lot of dissolved minerals (as different to soft water having a low dissolved mineral content). High levels of calcium (Ca2+) and magnesium (Mg2+) ions are present in hard water. Water hardness scale important to understand the topic.
- Concentration of multivalent cations is the definition of water hardness. Cations (metal ions) with a charge higher than 1+, primarily 2+, are referred to as multivalent cations. The cations Ca2+ and Mg2+ are among these.
- These ions leak into water supplies from minerals (like limestone) in aquifers. (An aquifer is a body/Part of rock and/or sediment that holds groundwater.)
- Calcite and gypsum are two common minerals that contain calcium. Dolomite, a common magnesium mineral that also includes calcium, is a good example.
- Because they have few ions, rainwater and distilled water are soft. Since the water in wells comes in close contact with minerals that can dissolve, hard water is frequently found there.
- Lakes, rivers, and streams’ surface water is typically soft with very little hardness.
- Although hard water hardly stances a human health risk, it could be a cause of major plumbing issues in both home and commercial settings.
- Water hardness is a simple problem, causing anything from the lack of suds development in bathing and laundry applications to costly error or malfunctions in boilers, cooling towers, and other water-handling machinery.
The formation/dissolution of calcium carbonate scales is described by the following equilibrium reaction:
CaCO3 + CO2 + H2O = 2HCO3– + Ca2+
Calcium and magnesium ions can be simply removed by the ion exchange procedure.
General guidelines for classification of waters :
- Measures of Waters
| Sr. No. | Calcium carbonate Concentration | Type of Waters |
| 1 | 0.00 to 60 mg/L | soft water |
| 2 | 61 to 120 mg/L | moderately hard water |
| 3 | 121 to 180 mg/L | hard |
| 4 | More than 180 mg/L | very hard |
Types of Hardness of Water
Water hardness can be divided into two categories:
- Temporary Hardness
- Permanent Hardness
Temporary Hardness of Water:
- Water becomes temporarily hard due to magnesium and calcium carbonates present. In this instance, boiling the water will get rid of the hardness.
- When water is heated to a boil, the soluble Mg(HCO3)2 salts are changed to the insoluble Mg(OH)2, which precipitates out and needs to be removed. The water we receive after filtering is soft water.
Permanent Hardness of Water:
- It will refer to permanent hardness when the presence of soluble magnesium (Mg) and calcium (Ca) salts in the form of chlorides and sulphides in water make it as permanent hardness since boiling cannot dissolve this hardness.
- By adding washing soda to the water, it can soften it up. When washing soda combines with the sulphide and chloride compounds of calcium and magnesium, insoluble carbonates are created, resulting in the transformation of hard water into soft water.
Disadvantages of Hardness of Waters
- Wastage of soap
- Wastage of fuel
- Formation of scales on metallic boilers.
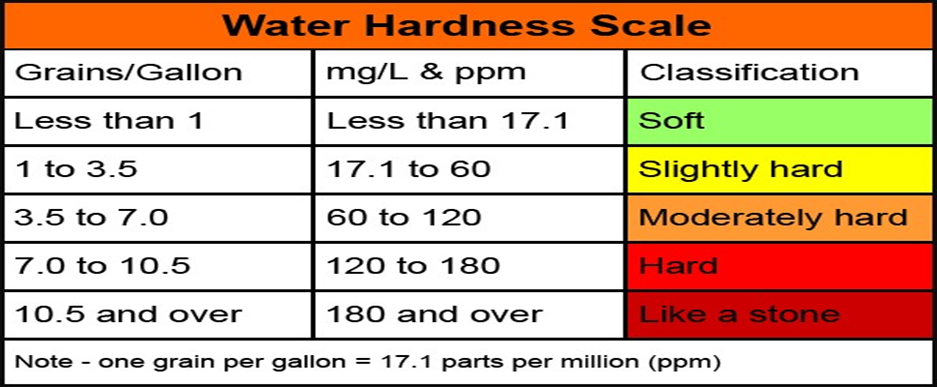
Water Hardness Scale shown as below,
Water softening:
It the process of removing the dissolved calcium and magnesium salts that cause hardness in water.
Water softening is achieved :
- By adding chemicals that form insoluble precipitates or
- By ion exchange.
By adding chemicals:
- Ammonia, Borax (Sodium Tetraborate), calcium hydroxide Ca(OH)2 (slaked lime), or trisodium phosphate are some of the chemicals used for softening on a modest scale, typically in conjunction with sodium carbonate (soda ash).
- To get rid of the precipitates, sedimentation and filtration must come after the lime-soda method of water softening.
- By adding just enough lime to precipitate the calcium as carbonate and the magnesium as hydroxide, and then sodium carbonate to eliminate the leftover calcium salts, water can be chemically softened on a massive scale.
By ion exchange:
- A typical industrial technique for softening water is ion exchange. By passing the water through columns of a natural or artificial resin that exchanges sodium ions (Na) for calcium (Ca) and magnesium (Mg) ions, the goal is achieved.
- After some time of use, calcium and magnesium start to show up in the water leaving the column.
- At that time, the column must be regenerated by slowly pouring a concentrated solution of common salt through it; the surplus sodium ions relocate the ions that cause the hardness so that, after being flushed with water, the bed of exchanger is once again ready for use.
- Initially, natural alumino silicates were utilised as the exchangers for this function, but later, synthetic resins took their place.
- The majority of home water softeners operate similarly and contain zeolite or another type of ion-exchange resin in a tank that is directly linked to the water system.

- A piece of equipment called a water softener is used to eliminate or reduce water hardness. The ion exchange process happens within this softener.
- Ion exchange resin is the main material found inside a water softener. Essentially an insoluble matrix, this resin often takes the form of tiny (1-2 mm in diameter) beads.
- The substance contains highly developed surface pores that serve as locations for ions that can be easily trapped and released. The word “ion-exchange” refers to the process of trapping ions while simultaneously releasing other ions.
- Ion exchange resins can be produced to prefer one or possibly more specific types of ions. These resins often have cross-linked polystyrene as their basis material.

People also search Similar Topic: Water Hardness Scale, Water hardness chart, Water hardness scale ppm, water hardness scale for water softener
Read More:
- Types of Autoclaves
- LAL test
- Water hardness scale
- LAL test Procedure
- TLC & HPTLC
- GxP in Pharmaceuticals
- Types of chromatography
- CIP and SIP in Pharmaceuticals
- Positive control and negative control in Microbiology
- Types of biological indicators for sterilization
- Sterility Assurance Level (SAL)
- D value and Z values in Microbiology
- SOP on “Operation and Cleaning of Vertical Autoclave”
- Vacuum Leak Test, acceptance criteria and pharmaceutical applications
- Tablets Hardness Testing
- Quality Assurance
- Analytical Development or Quality Control Formulation Development
- Microbiology
- Health Topic
Reference:
- Water softening – Wikipedia
- Water Hardness and Water softening
- https://www.luminoruv.com/education/softening/
- water softening | Definition, Process, & Facts | Britannica
