Free Energy, Endergonic, and Exergonic Reactions
Introduction:
- Free energy, endergonic, and exergonic reactions are fundamental concepts in chemistry and biology.
- They provide insights into the energy changes that occur during chemical reactions, which are crucial for understanding how living organisms function and interact with their environment.
- In this comprehensive guide, we’ll delve into these concepts, explaining what free energy is, and how endergonic and exergonic reactions play essential roles in various biological processes.
1. Free Energy :
- Free energy, denoted as G, is a critical concept in thermodynamics and biochemistry. It represents the energy available in a system to do work at constant temperature and pressure. Free energy is a measure of a system’s capacity to perform useful work, such as driving chemical reactions, maintaining cellular processes, or even powering your body’s movements.
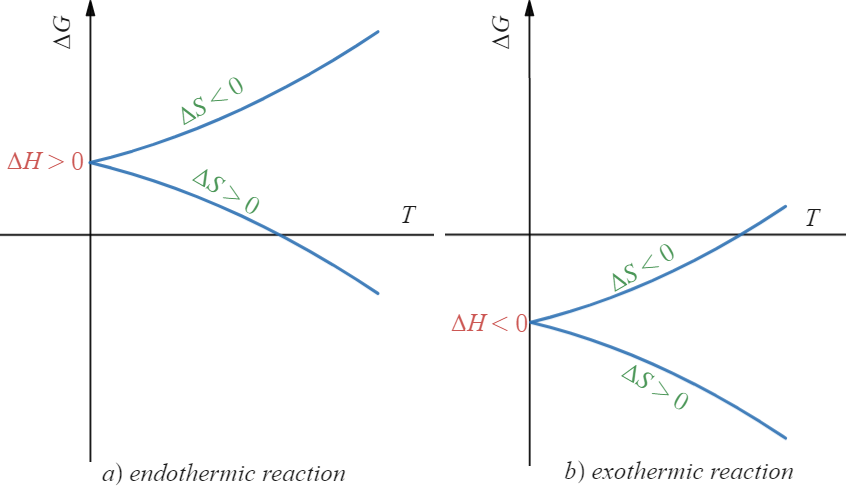
The Gibbs free energy equation,
ΔG = ΔH – TΔS,
- provides a quantitative measure of free energy changes during chemical reactions. Here’s a breakdown of the equation:
– ΔG: Change in free energy
– ΔH: Change in enthalpy (total energy of a system)
– T: Temperature in Kelvin
– ΔS: Change in entropy (a measure of disorder or randomness)
- In a nutshell, a negative ΔG indicates that a reaction is exergonic, meaning it releases free energy, while a positive ΔG signifies an endergonic reaction, requiring an input of energy.
2. Endergonic Reactions
- Endergonic reactions are a type of chemical reaction that absorbs free energy from their surroundings.
- These reactions are not spontaneous and require an input of energy to proceed. In other words, the products of endergonic reactions have more free energy than the reactants, resulting in a positive ΔG.
- Endergonic reactions are prevalent in biological systems, where energy input is essential for maintaining life processes. An excellent example is photosynthesis, the process by which plants convert sunlight into chemical energy.
- In this complex series of reactions, carbon dioxide and water are transformed into glucose and oxygen, storing energy in the form of chemical bonds. The energy for photosynthesis is provided by sunlight, making it an endergonic process.
- Another critical example of an endergonic reaction is the synthesis of adenosine triphosphate (ATP) in cells. ATP is often referred to as the “energy currency” of cells because it stores and transfers energy within biological systems.
- Cells use endergonic reactions to produce ATP from adenosine diphosphate (ADP) and inorganic phosphate (Pi). The energy needed for this process comes from exergonic reactions elsewhere in the cell, such as the breakdown of glucose during cellular respiration.
- Overall, endergonic reactions play a vital role in maintaining the energy balance within biological systems and ensuring that cells have the energy needed to carry out their functions.
3. Exergonic Reactions:
- Exergonic reactions are the opposite of endergonic reactions. These reactions release free energy and are spontaneous under certain conditions. In an exergonic reaction, the products have lower free energy than the reactants, resulting in a negative ΔG.
- Exergonic reactions are essential for powering various biological processes. One of the most well-known exergonic reactions is cellular respiration, where glucose and oxygen are broken down to produce carbon dioxide, water, and ATP.
- This process releases a significant amount of free energy, which cells can use to perform various tasks, such as muscle contraction, active transport, and DNA replication.
- Another example of an exergonic reaction is the hydrolysis of ATP into ADP and Pi.
- This reaction is frequently coupled with endergonic reactions to provide the necessary energy input.
- For instance, during muscle contraction, the energy released by the hydrolysis of ATP fuels the mechanical work of muscle fibers, enabling movement.
- Exergonic reactions are also crucial for maintaining temperature and chemical balance in living organisms. They release heat as a byproduct, helping to regulate body temperature.
- Additionally, exergonic reactions are involved in the breakdown of waste products and toxins in the body, ensuring proper detoxification.
Picture Credit@Wikipedia
Conclusion :
- In summary, free energy, endergonic, and exergonic reactions are fundamental concepts that shed light on the energy changes that occur during chemical reactions.
- Free energy, as measured by ΔG, represents a system’s ability to do work, with negative values indicating exergonic reactions and positive values indicating endergonic reactions.
- Endergonic reactions absorb energy and are vital for processes like photosynthesis and ATP synthesis, while exergonic reactions release energy and power activities such as cellular respiration and muscle contraction.
- Understanding these concepts is crucial for unraveling the mysteries of life processes and energy flow in biological systems.
Read More:
- The 6M Method in Pharmaceutical Laboratory Investigations
- 5 whys method in pharmaceutical industry Different types of dosage forms in Pharmaceutical Industries
- What are the Different types of Modified Dosage Forms?
- what is an extractables and leachables testing test in pharmaceuticals ?
- What is difference between FBP and FBD in Pharmaceutical Manufacturing
