What is the 6M Method in Pharmaceutical Laboratory Investigations ?
6M Method in Pharmaceutical Laboratory Investigations is used for cause and effect analysis in Lab. investigation in Pharma production. It includes Manpower, Method, Machine, Material, Milieu and Measurement.
Introduction :
- In the realm of pharmaceutical laboratories, conducting thorough investigations and root cause analysis is crucial for ensuring product quality, safety, and efficacy.
- One widely adopted approach in this domain is the 6M method.
- This article aims to provide a comprehensive guide to the 6M method, its significance, and its application in laboratory investigations within the pharmaceutical industry.
- By understanding and implementing this method effectively, pharmaceutical professionals can enhance their problem-solving capabilities, optimize processes, and uphold the highest standards of quality control.
Understanding the 6M Method:
- The 6M method, also known as the 6Ms, is a structured approach used in pharmaceutical laboratory investigations to identify potential sources of variation or causes that may contribute to issues or problems.
- The six factors encompassed by the 6M method are manpower, method, machine, material, measurement, and mother nature (environment).
- Each of these factors plays a significant role in laboratory investigations and must be thoroughly evaluated to ensure accurate analysis and effective problem resolution.
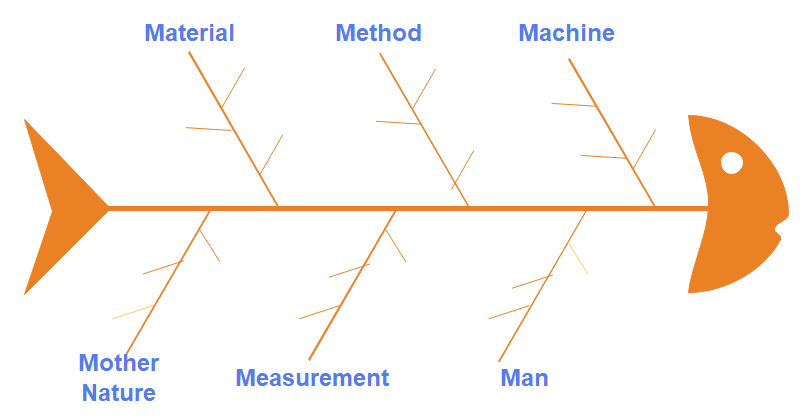
@credit to: https://www.edrawsoft.com
1. Manpower: The Human Element
- The first factor of the 6M method, manpower, focuses on human operators involved in laboratory investigations.
- This section delves into the importance of well-trained personnel, adherence to standard operating procedures, and the impact of human error on investigation outcomes.
- Additionally, it explores strategies for training, skill development, and ongoing performance monitoring to minimize human-related variations in pharmaceutical laboratory investigations.
2. Method: Procedures and Protocols
- The method factor of the its emphasizes the procedures, methods, and protocols used in laboratory investigations.
- This section discusses the significance of following established methodologies, the potential impact of deviations, and the importance of documentation.
- It also highlights the need for continuous improvement and validation of testing methodologies to ensure reliable and accurate results.
3. Machine: Equipment and Instrumentation:
- The machine factor focuses on the equipment, instruments, and machinery employed in pharmaceutical laboratory investigations.
- This section explores the significance of proper calibration, routine maintenance, and adherence to equipment specifications.
- It also discusses the potential impact of equipment malfunctions or inadequacies on the investigation process and the importance of prompt resolution to maintain data integrity.
4. Material: Quality and Integrity :
- Material refers to the raw materials, reagents, and samples used in laboratory investigations.
- This section emphasizes the importance of sourcing high-quality materials, ensuring their integrity and traceability, and preventing contamination or degradation.
- It explores strategies for robust supplier qualification, storage and handling practices, and effective inventory management to mitigate potential material-related issues.
5. Measurement: Accuracy and Reliability :
- Measurement encompasses the accuracy and reliability of the instruments, tools, and techniques used in laboratory investigations.
- This section discusses the importance of proper calibration, validation, and routine performance checks to ensure precise measurements.
- It also explores the significance of data integrity, appropriate statistical analysis, and the use of reliable reference standards.
6. Mother Nature: Environmental Considerations :
- The final factor of the 6M method, mother nature (environment), focuses on the impact of external factors on laboratory investigations.
- This section explores the influence of environmental conditions such as temperature, humidity, lighting, and cleanliness.
- It discusses the importance of adequate controls, monitoring, and mitigation strategies to minimize variations caused by environmental factors.
Conclusion of 6M Method in Pharmaceutical Laboratory Investigations:
- It provides a structured approach for investigating and resolving issues in pharmaceutical laboratories.
- By thoroughly evaluating the factors of manpower, method, machine, material, measurement , and mother nature, professionals in the pharmaceutical industry can identify potential sources of variation, implement effective corrective actions, and enhance the overall quality control process.
- Adhering to the principles of the 6M method promotes consistent and reliable laboratory investigations, enabling pharmaceutical companies to ensure the safety, efficacy, and quality of their products.
- By continually improving their understanding and application of the 6M method, pharmaceutical professionals can contribute to the advancement of laboratory practices and maintain the highest standards of excellence within the industry.
Read More:
- Classification of Tablet Coating
- HEPA filter in the pharmaceutical industry
- Buccal and Sublingual Tablets
- Type of Tablets
- Compressibility index and Hausner ratio
- Tapped density and Bulk density
- 6M method for cause and effect analysis
- 6M method for root cause analysis
- Quality Assurance
- Analytical Development or Quality Control Formulation Development
- Microbiology
- Health Topic
