pH Meter Principle:
To determine and measure acidity or alkalinity of solutions pH meter is used. ph meter working principle is mainly based on concentration of H+ ions.
- The concentration of H+ ions in the acidic solution is high. The H+ ion moves in close proximity to the glass membrane of the sensitive glass bulb when the pH probe is submerged in an acidic solution (external side of the bulb).
- Inside the bulb, which is filled with buffer solution with a pH of neutral, a similar reaction takes place.
- The quantity of hydrogen ions in this neutral buffer solution is constant. Additionally, the H+ ions in the bulb travel in close proximity to the glass membrane (internal side of the bulb).
- Which leads to the difference in the concentration of hydrogen ion (H+ ions) or degree of hydrogen ion activity across the membrane causing a difference in the potential (voltage).
- When the provided solution is acidic and has a pH lower than 7 when the hydrogen ion concentration inside the glass bulb is lower than the outside solution (test solution).
- An increase in the solution’s acidity results in a higher concentration of hydrogen ions (H+), which raises the voltage. The pH reading in pH meter falls as a result of the higher voltage.
- The pH is equal to 7 when the concentration of hydrogen ions across the membrane is the same, which is referred to as neutral pH.
- The provided solution is alkaline and has a pH more than 7 if the hydrogen ion concentration inside the bulb is greater than that of the surrounding solution.
- The pH metre calculates pH based on the voltage difference between the two electrodes as per the Nernst equation.
- Hydrogen ions in the test solution swap places with other positively charged ions on the glass bulb upon immersion in the solution under test, creating an electrochemical potential across the bulb.
- The difference in electric potential between the two electrodes is picked up by the amplifier. The pH unit refers to the contrast between these potentials.
Basic of pH Meter or Logic behind pH Meter Principle:
Measuring of the pH required assembly, or sensor, consists of two primary parts:
- Measuring electrode: The measuring electrode is occasionally called the glass electrode, and is also denoted as a membrane or active electrode.
- Reference electrode: The reference electrode is also denoted as a standard electrode.
There are two half-cells, or electrode, potentials used to measure pH. The pH-sensitive glass measuring electrode is housed in one half of the cell, and the reference electrode is in the other. A pH sensor needs two half-cell potentials to complete a circuit, much like a battery does.
The mathematical equation for this is:
E = Em-Er
Where,
- Em = the electrode potential -measuring electrode
- Er = the electrode potential – reference electrode.
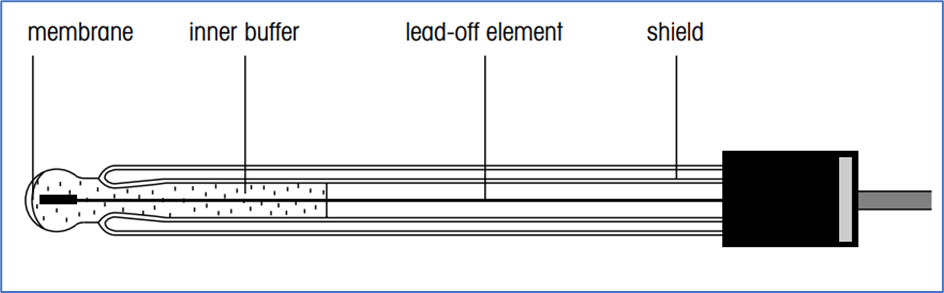
Glass electrodes or Measuring electrode:
- When an aqueous measuring solution and a pH glass electrode come into contact, a “gel layer” forms on the pH-sensitive glass membrane.
- A similar “gel layer” forms inside the glass membrane when it comes into contact with a specific buffer solution (the inner buffer).
- Depending on the pH of the measured solution, the H+ ions either diffuse into or out of the gel layer.
- In an alkaline solution, the H+ ions diffuse out and leave a negative charge on the gel layer’s outer surface.
- The potential at the inner surface of the membrane is constant during the measurement because the glass electrode contains an internal buffer with a constant pH value.
- The difference between the inner and outside charges leads to the overall membrane potential.
Equation: Eel = E0– S (pHa – pHi)
- Eel = electrode potential
- E0 = zero potential
- S = slope (mV per pH unit)
- pHi = stand for pH value of the internal buffer
- pHa = stand for pH value of the measured solution
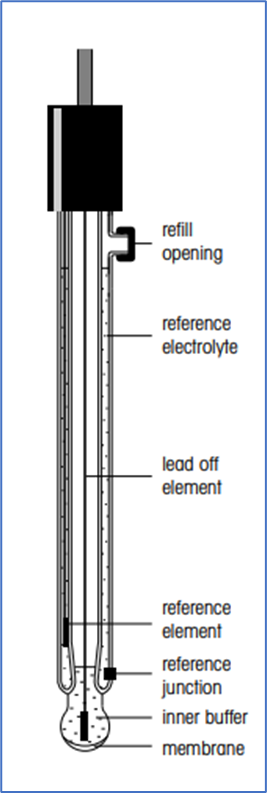
Reference electrodes
- A measuring electrode and a reference electrode, both of which are submerged in the same solution, make up the entire pH measurement circuit.
- The reference electrode needs to have a fixed, stable voltage that is independent of the measured solution in order to get a precise pH value.
- A reference element that is submerged in a specific electrolyte makes up each reference electrode.
- The measured solution must be in contact with this electrolyte. Most frequently, this contact happens through a porous ceramic junction.
- Only two reference systems: mercury/calomel and silver/silver chloride—as well as several of their variants have reached practical significance. Due to environmental concern, the mercury electrode is now hardly ever utilised.
- The reference electrolyte and the reference element (such as silver/silver chloride) determine the potential of the reference electrode system.
- Here, a high ion concentration in the reference electrolyte which results in a low electrical resistance is key.

Three-in-one electrodes (Glass + Reference +Temperature Sensor)
- The addition of a temperature sensor to the pH combination electrode is a new innovation.
- Temperature-compensated readings can be conveniently taken with a single probe by placing the temperature sensor in the same body as the pH and reference elements.
Read : Process of Regeneration of pH Electrode, ph meter principle
Limitation of pH meter:
pH meters are widely used to measure the acidity or alkalinity of a solution. While they are generally reliable and accurate, there are a few limitations associated with their use. Here are some limitations of pH meters:
- Calibration: pH meters require regular calibration using standard buffer solutions to maintain accuracy. If the meter is not calibrated properly or if the calibration is outdated, it can lead to inaccurate pH measurements.
- Electrode performance: pH meters utilize a glass electrode to measure pH. Over time, the electrode can deteriorate or get contaminated, which can affect the accuracy of the measurements. Regular electrode maintenance and replacement are necessary to ensure reliable results.
- Temperature effects: pH measurements are temperature-dependent, and most pH meters have built-in temperature compensation features. However, extreme temperatures or rapid temperature changes can still affect the accuracy of pH readings. It is important to use the meter within its specified temperature range and allow sufficient time for temperature equilibration.
- Interference: Certain substances or ions present in the sample can interfere with pH measurements. For example, highly concentrated solutions, colloidal suspensions, or substances with high ionic strength can cause deviations from the true pH value. In such cases, additional sample preparation or alternative measurement techniques may be required.
- Sample characteristics: pH meters are designed to measure the pH of aqueous solutions. They may not be suitable for measuring the pH of non-aqueous solutions or samples with high viscosity or solids content. The presence of oils, fats, or other non-aqueous components can affect the accuracy of pH readings.
- Electrode drift: pH electrodes can experience drift over time, where the readings gradually change even in the absence of any pH changes in the sample. This drift can lead to inaccurate measurements if not accounted for by periodic recalibration or electrode replacement.
- Maintenance and storage: Proper maintenance and storage of pH meters are crucial to ensure their longevity and accuracy. Improper storage or neglecting regular maintenance can result in electrode damage, reduced sensitivity, or inconsistent readings.
It’s important to understand these limitations and take appropriate measures to mitigate their impact on pH measurements or ph meter principle. Regular calibration, electrode maintenance, adherence to operating conditions, and awareness of potential interferences are essential for obtaining reliable pH data
Advantages of pH Meters:
- Accuracy: pH meters are generally highly accurate and provide precise measurements of pH values. They offer a direct and quantitative assessment of the acidity or alkalinity of a solution.
- Wide Range of Measurement: pH meters can measure pH across a broad range, typically from pH 0 to pH 14. This versatility makes them suitable for a wide range of applications, from testing the pH of soil and water to monitoring the pH of biological samples and industrial processes.
- Quick and Real-time Measurements: pH meters provide rapid measurements, allowing for real-time monitoring of pH changes. This is particularly useful in dynamic processes or experiments where immediate pH adjustments may be necessary.
- Ease of Use: pH meters are generally user-friendly and straightforward to operate. Modern pH meters often come with intuitive interfaces and digital displays, making them accessible even to individuals with limited technical expertise.
- Portable and Handheld Options: Many pH meters are designed as portable and handheld devices, allowing for convenient on-site testing. This portability makes them useful in fieldwork, environmental monitoring, and other applications where measurements need to be taken outside the laboratory.
Disadvantages of pH Meters:
- Cost: High-quality pH meters can be relatively expensive, especially those with advanced features and capabilities. The initial investment cost, along with the cost of calibration solutions and electrode maintenance, may be a barrier for some users or organizations.
- Calibration and Maintenance: pH meters require regular calibration using standard buffer solutions to maintain accuracy. Calibration can be time-consuming and may require additional equipment and solutions. Furthermore, pH electrodes require proper maintenance, storage, and occasional replacement, which adds to the overall cost and effort.
- Sensitivity to Environmental Factors: pH meters can be sensitive to factors such as temperature, humidity, and electrostatic interference. It is important to use pH meters within their specified operating conditions and take necessary precautions to minimize environmental effects.
- Limited Application to Aqueous Solutions: pH meters are primarily designed for measuring the pH of aqueous solutions. They may not be suitable for non-aqueous samples or those with high viscosity, solids content, or complex matrices. In such cases, alternative measurement techniques may be necessary.
- Interference: Certain substances or ions present in the sample can interfere with pH measurements, leading to inaccurate readings. It is important to be aware of potential interferences and take appropriate measures to minimize their impact, such as sample preparation or using specialized electrodes.
What is Effect of temperature on pH measurement?
- Temperature has a significant effect on pH measurements. The pH value of a solution is temperature-dependent, meaning that changes in temperature can affect the measured pH value. Here’s how temperature influences pH measurements:
- Electrode Sensitivity: pH meters use a glass electrode to measure pH, which consists of a pH-sensitive glass membrane. The sensitivity of the glass electrode to hydrogen ions (H+) changes with temperature. As temperature increases, the sensitivity of the electrode decreases, leading to lower pH readings, and vice versa. Therefore, it is essential to consider temperature compensation when measuring pH.
- pH Scale Variation: The pH scale itself is temperature-dependent. At different temperatures, the dissociation constants of water and acidic/basic substances change, causing variations in the pH scale. The standard pH scale is defined at 25 degrees Celsius, and deviations from this temperature can introduce errors in pH measurements.
- Ionization of Water: The ionization of water molecules into hydrogen ions (H+) and hydroxide ions (OH-) is temperature-dependent. As temperature rises, the concentration of hydrogen ions increases, resulting in a decrease in pH. Conversely, as temperature decreases, the concentration of hydroxide ions increases, leading to an increase in pH.
- Temperature Compensation: To account for the temperature effect on pH measurements, pH meters often include automatic temperature compensation (ATC) features. ATC adjusts the pH reading based on the measured temperature, using algorithms and predetermined temperature coefficients. This compensates for the temperature-induced changes in electrode sensitivity and the pH scale, providing more accurate pH values.
- Electrode Performance: Extreme temperatures can affect the performance and lifespan of the pH electrode. Exposing the electrode to very high or low temperatures may lead to electrode drift, reduced sensitivity, or even physical damage. It is important to use pH meters within their specified temperature range and allow sufficient time for temperature equilibration before taking measurements.
To ensure accurate pH measurements, it is crucial to calibrate the pH meter using standard buffer solutions at the intended operating temperature. Additionally, employing temperature compensation features and maintaining the temperature stability of the measurement environment can help minimize temperature-related errors in pH measurements.
Precautions of pH meter:
When using a pH meter, it is important to take certain precautions to ensure accurate and reliable measurements. Here are some key precautions to consider:
- Calibration: Regularly calibrate the pH meter using standard buffer solutions. Calibrating before each use or at least daily is recommended. Follow the manufacturer’s instructions for calibration procedures and use fresh buffer solutions within their expiration dates.
- Electrode Maintenance: Properly clean and maintain the pH electrode. Rinse the electrode with distilled water before and after each measurement to remove any residue or contaminants. If necessary, use a suitable cleaning solution recommended by the manufacturer. Store the electrode in a storage solution or cap filled with electrode storage solution when not in use to prevent drying out and maintain its performance.
- Temperature Compensation: Use pH meters equipped with automatic temperature compensation (ATC) features. Ensure that the temperature sensor is in close proximity to the sample being measured for accurate temperature readings. Allow sufficient time for temperature equilibration before taking pH measurements.
- Sample Handling: Handle samples carefully and avoid cross-contamination. Clean the sample container thoroughly or use disposable containers to prevent contamination from previous samples. Avoid touching the pH electrode directly with fingers or other objects, as this can introduce contaminants or damage the electrode.
- Stirring and Mixing: For accurate measurements, stir or mix the sample gently to ensure uniform distribution of ions and temperature throughout the solution. This helps to achieve a stable and representative pH reading.
- Avoid Extreme pH Ranges: pH meters are typically designed to measure within a specific pH range. Avoid measuring solutions that are too acidic or too alkaline for the specified range of the pH meter, as this can lead to inaccurate measurements or damage to the electrode.
- Environmental Factors: Ensure that the pH meter and samples are at the appropriate temperature and humidity levels specified by the manufacturer. Minimize exposure to extreme temperatures, direct sunlight, moisture, and electrostatic interference, as these factors can affect the accuracy and performance of the pH meter.
- Regular Maintenance and Storage: Follow the manufacturer’s guidelines for regular maintenance, electrode replacement, and storage of the pH meter. Clean the meter’s external surfaces as needed, and store it in a clean, dry, and protected environment when not in use.
By following these precautions, you can enhance the accuracy and longevity of your pH meter, ensuring reliable pH measurements in various applications. Always refer to the specific guidelines provided by the manufacturer of pH meter for precise instructions and recommendations.
Reference:
- Mettler Toledo
- pH meter
- FAQ on pH Meters & ph meter principle
Read More:
- Difference Between Isocratic and Gradient Elution
- Calibration of HPLC
- pH meter principle and application
- Calibration and Operation of pH meter
- Gas Chromatography Columns
- Why is 70% the More Effective Concentration of Isopropyl Alcohol for Disinfection?
- Difference between Incidence and Deviation
- Differential Scanning Calorimetry (DSC)
- Paper Chromatography
- ph meter principle
- Thin Layer Chromatography
- Difference Between Thin Layer and Paper Chromatography
- Type of Glass container used in Pharmaceuticals Fume Hood Type of HPLC Column
- Type of Capsules
