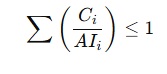Nitrosamine Impurities in Pharmaceuticals
What are Nitrosamines?
Nitrosamine Impurities: Nitrosamines are a class of organic compounds characterized by a nitroso group (–N=O) bonded to a secondary or tertiary amine (NR₂).
Common examples include N-nitrosodimethylamine (NDMA) and N-nitrosodiethylamine (NDEA). While nitrosamines have industrial applications, their unintended presence in medicines is problematic because many are classified as probable human carcinogens.
Common Nitrosamine Impurities
Several nitrosamines have been identified in pharmaceuticals, including:
- NDMA (N-Nitrosodimethylamine)
- NDEA (N-Nitrosodiethylamine)
- NMBA (N-Nitroso-N-methyl-4-aminobutyric acid)
- NDIPA (N-Nitrosodiisopropylamine)
- NMPA (N-Nitrosomethylphenylamine)
These have been detected in various drug classes like sartans (antihypertensives), ranitidine (acid reducer), and metformin (antidiabetic agent).
How Do Nitrosamines End Up in Pharmaceuticals?
Nitrosamine impurities can enter pharmaceutical products through several pathways:
- Manufacturing Processes: Nitrosamines may form during drug synthesis if certain solvents, catalysts, or reagents are used. For instance, sodium nitrite in the presence of secondary or tertiary amines under acidic conditions can lead to nitrosamine formation.
- Contaminated Raw Materials: Impurities in starting materials or intermediates sourced from suppliers can introduce nitrosamines.
- Recycled Solvents: Reuse of solvents like dimethylformamide (DMF) can inadvertently introduce or generate nitrosamine impurities if not properly controlled.
- Packaging and Storage: Nitrosamines can form during storage due to interactions between drug formulations and packaging materials or environmental factors.
The complexity of pharmaceutical supply chains and variability in manufacturing practices globally make it challenging to eliminate these impurities entirely.
Why are Nitrosamine Impurities a concern?
- Some nitroso compounds are mutagenic or carcinogenic.
- Their presence in pharmaceutical products, even in trace amounts, is tightly regulated.
- They can form during synthesis, degradation, or storage of drug substances and intermediates.
Examples of Nitroso Impurities
| Nitroso Impurity | Type | Source or Context |
|---|---|---|
| Nitrosamines | Secondary nitroso compounds | Formed from amines + nitrites |
| Nitrosoaniline | Aromatic nitroso compound | Byproduct in dye intermediates |
| N-Methyl-N-nitrosoaniline | Nitrosamine | Intermediate Degradation product |
| N-Nitrosodimethylamine (NDMA) | Nitrosamine | Found in ranitidine, metformin, sartans |
| N-Nitrosodiethylamine (NDEA) | Nitrosamine | Found in valsartan |
| Nitrosophenols | Phenol derivatives | May occur via oxidative degradation |
| N-Nitrosomorpholine | Cyclic nitrosamine | Found as impurity or degradation product |
| N-Nitrosopyrrolidine | Cyclic nitrosamine | Possible byproduct from pyrrolidine use |
What are Nitrosating agents?
Nitrosating agents are chemical compounds that introduce a nitroso group (–NO) into other molecules, typically amines, to form nitrosamines. These agents are key contributors to the unintentional formation of nitrosamine impurities in pharmaceuticals.
Types of Amines and Why Are Secondary and Tertiary Amines Responsible for Nitrosamine Impurities?
Secondary and tertiary amines are key precursors in the formation of nitrosamine impurities due to their reactivity with nitrosating agents, leading to potentially carcinogenic nitrosamines.
Basic Chemistry Behind It:
Nitrosamines form via a nitrosation reaction involving:
- Primary Amines (R-NH₂): One alkyl or aryl group attached to nitrogen.
- Less likely to form stable nitrosamines, often forming unstable diazonium salts that decompose quickly.
- Examples: Methylamine, Ethylamine.
- Nitrosamine risk: Low.
- Secondary Amines (R₂NH): Two alkyl or aryl groups attached to nitrogen.
- High risk, readily react with nitrosating agents to form stable nitrosamines.
- Examples:
- Dimethylamine (DMA) → NDMA,
- Diethylamine (DEA) → NDEA,
- Morpholine → NMOR, Piperidine → NPIP.
- Nitrosamine risk: High.
- Tertiary Amines (R₃N): Three alkyl or aryl groups attached to nitrogen.
- Do not directly form nitrosamines but can degrade into secondary amines.
- Examples:
- Triethylamine (TEA),
- Trimethylamine (TMA),
- Diisopropylethylamine (DIPEA) → NDIPA via degradation.
- Nitrosamine risk: Moderate to High, indirectly.
Why are the Amines Problematic? or What are the Reason behind the Amines are more likely for Nitrosamine Impurities?
| Factor | Why It Contributes |
|---|---|
| Nucleophilicity of amines | Amines easily attack the electrophilic nitrosating species (like NO⁺). |
| Stability of nitrosamines | Once formed, nitrosamines are stable and persist in the product. |
| Presence of nitrites/nitrates | Common in reagents, water, or excipients — reacts with amines. |
| Reaction under mild conditions | Reaction can occur at room temperature and low pH (e.g., pH 3–5). |
Examples of Secondary and Tertiary Amines & Their Nitrosamine Products:
| Amine (Source) | Type | Drug/Product Context | Nitrosamine Formed |
|---|---|---|---|
| Dimethylamine (DMA) | Secondary amine | Degradation product of DMAc, DMF, or certain APIs | NDMA (N-Nitrosodimethylamine) |
| Diethylamine (DEA) | Secondary amine | Impurity or reagent in API synthesis | NDEA (N-Nitrosodiethylamine) |
| Isopropylamine | Secondary amine | Used in synthesis of some APIs | NDIPA (N-Nitrosodiisopropylamine) |
| Methylphenylamine | Secondary amine | Intermediate or impurity | NMPA (N-Nitrosomethylphenylamine) |
| Morpholine | Secondary amine | Solvent or stabilizer | NMOR (N-Nitrosomorpholine) |
| Pyrrolidine | Secondary amine | Intermediate or degradation product | NPYR (N-Nitrosopyrrolidine) |
| Piperidine | Secondary amine | Common ring in drug scaffolds (e.g., antihistamines) | NPIP (N-Nitrosopiperidine) |
| Triethylamine (TEA) | Tertiary amine | Base or pH adjuster in synthesis | Forms DEA, which can form NDEA |
| Tetramethylammonium hydroxide | Tertiary amine | Used in analytical and synthetic processes | Degrades to DMA → NDMA |
| Tributylamine | Tertiary amine | Catalyst or base in synthesis | Can degrade into secondary butylamines |
| 2° amine-containing APIs | Secondary amine | e.g., Ranitidine, Nizatidine | Degrade into NDMA under heat/acid |
| API intermediates with amino groups | Secondary amine | Varies (e.g., sartans, anti-diabetics) | Form specific nitrosamines based on structure |
Where Do These Amines Come From?
| Source Type | Examples | Risk Contribution |
|---|---|---|
| Solvents | DMF (Dimethylformamide), DMAc (Dimethylacetamide), NMP, TEA | May contain or degrade to secondary amines like DMA |
| Reagents / Catalysts | Diethylamine, isopropylamine, morpholine, pyrrolidine | Direct sources of secondary amines |
| API / Intermediates | Drug substances with amine functionalities (e.g., piperidine, morpholine rings) | Can participate in nitrosation reactions or degrade to reactive amines |
| Excipients | Magnesium stearate, talc, microcrystalline cellulose (if contaminated) | May contain nitrites or residual amines as impurities |
| Water | Recycled or industrial-grade water | May contain trace nitrites and other ions that support nitrosation |
| Cleaning Agents | Triethylamine-based cleaning solvents used in GMP facilities | Residual amines left on equipment can react during production |
| Degradation Products | From drug substance or excipients during shelf-life | e.g., Ranitidine decomposes to NDMA |
| Packaging Materials | Blister films, inks, adhesives, nitrocellulose-based coatings | Can release amines or nitrites over time or under heat/humidity |
| Gaskets & Liners | Rubber stoppers, laminated seals, container closures | May contain amine-based antioxidants, accelerators, or residuals |
| Ink and Printing | Nitrocellulose inks on foil or label | Can release NOx compounds and react with amines nearby |
Extractables and leachable (E&L) studies are critical when assessing nitrosamine risk.
Example in Real Drugs
| Drug | Nitrosamine Found | Amines Involved |
|---|---|---|
| Valsartan | NDMA, NDEA | DMA (from solvent), DEA (impurity) |
| Ranitidine | NDMA | Secondary amine in API itself |
| Metformin | NDMA | Possibly from nitrite contamination |
| Irbesartan | NDIPA | Isopropylamine used in synthesis |
| Losartan | NMBA | Byproduct of butyric acid derivatives |
Step-by-Step: Nitrosamine Impurity Calculation
- Determine the Acceptable Intake (AI) LimitRegulatory agencies like the EMA, FDA, and ICH provide Acceptable Intake (AI) values for individual nitrosamines:
Nitrosamine AI Limit (ng/day) Source NDMA (N-Nitrosodimethylamine) 96 ng/day EMA, FDA NDEA (N-Nitrosodiethylamine) 26.5 ng/day EMA, FDA NMBA, NDIPA, NEIPA, etc. Varies Case-by-case - Calculate Nitrosamine Concentration in Drug (ng/g or ng/tablet): Use validated analytical methods (like LC-MS/MS or GC-MS) to determine how much nitrosamine is present in the sample.Example: If you test 1 tablet (500 mg) and detect 48 ng of NDMA, the nitrosamine content is:NDMA concentration = 48 ng per 500 mg tablet
- Compare with Acceptable Intake (AI) LimitTo check if the detected level is within the safe limit, use the formula:Daily Intake (ng/day) = Nitrosamine content per unit × Daily dose unitsExample: If a patient takes 2 tablets per day:Daily NDMA Intake = 48 ng × 2 = 96 ng/day
This matches the AI limit for NDMA → Acceptable.
4. Calculate Maximum Allowable Nitrosamine Content per Unit: If you need to find how much nitrosamine is allowed per tablet, use:
Max per unit = AI Limit (ng/day) / Max daily dose units
Example for NDMA:
- AI limit = 96 ng/day
- Daily dose = 2 tablets/dayMax NDMA per tablet=96/2 = 48 ng/Tablet
5. For Multiple Nitrosamines – Use a Combined Risk Approach
If more than one nitrosamine is present:
Where:
- Ci = concentration of nitrosamine i
- AIi = acceptable intake of nitrosamine i
This is known as the “Sum of Ratios” approach per ICH M7 and EMA guidelines
Use of PDE-Based Calculation in Nitrosoamine Imp:
Where: PDE: Permitted Daily Exposure (PDE)
Example with respect to Formulation :
Example 1: Valsartan (linked to NDMA)
- Nitrosamine: NDMA
- PDE: 0.096 µg/day (96 ng/day)
- Max daily dose of Valsartan: 320 mg
Limit=(0.096×1000)/320=0.3 ppm
NDMA limit in Valsartan = 0.3 ppm
Example 2: Metformin (linked to NDMA)
- Nitrosamine: NDMA
- PDE: 0.096 µg/day
- Max daily dose of Metformin: 2000 mg (2 g)
Limit=(0.096×1000)/2000=0.048 ppm
NDMA limit in Metformin = 0.048 ppm
Example 3: Losartan (linked to NMBA)
- Nitrosamine: NMBA (N-Nitroso-N-methyl-4-aminobutyric acid)
- PDE: 0.096 µg/day
- Max daily dose of Losartan: 100 mg
Limit=(0.096×1000)/100=0.96 ppm
NMBA limit in Losartan = 0.96 ppm
Example 4: Ranitidine (self-degrades to NDMA)
- Nitrosamine: NDMA
- PDE: 0.096 µg/day
- Max daily dose of Ranitidine: 300 mg
Limit=(0.096×1000)/300=0.32 ppm
NDMA limit in Ranitidine = 0.32 ppm
Example 5: Irbesartan (linked to NDIPA)
- Nitrosamine: NDIPA (N-Nitrosodiisopropylamine)
- PDE: ~0.0265 µg/day (based on structural alerts)
- Max daily dose of Irbesartan: 300 mg
Limit=(0.0265×1000)/300=0.0883 ppm
NDIPA limit in Irbesartan = ~0.088 ppm
Reference:
- GENERAL CHAPTER <1469> NITROSAMINE IMPURITIES
- Nitrosamine impurities | European Medicines Agency (EMA)
- Information Note Nitrosamine impurities
- HPLC Principle
- Nitrosamine Drug Substance-Related Impurities (NDSRIs)
- Nitrosamine
- Quality Assurance
- ICH
- Analytical Evaluation Threshold (AET) in Pharmaceutical
- Gas Chromatography Detector (Type of GC Detectors)