Gas Chromatography Detector
Below lists are the GC Detector types
- Flame ionization Detector(FID)
- Thermal conductivity Detector(TCD)
- Electron Capture Detector (ECD)
- Flame Thermionic Detector (FTD)
- Nitrogen-phosphorus Detector
- Flame photometric Detector (FPD)
- Photo-ionization Detector (PID)
- Electrolytic Conductivity Detector (ELCD)
- Barrier Discharge Ionization Detector (BID)
- Sulfur chemiluminescence Detector (SCD)
- Gas chromatography–mass spectrometry (GC-MS)
Related: Gas Chromatography Columns, Principle of HPLC
Type and Details of Gas chromatography Detectors
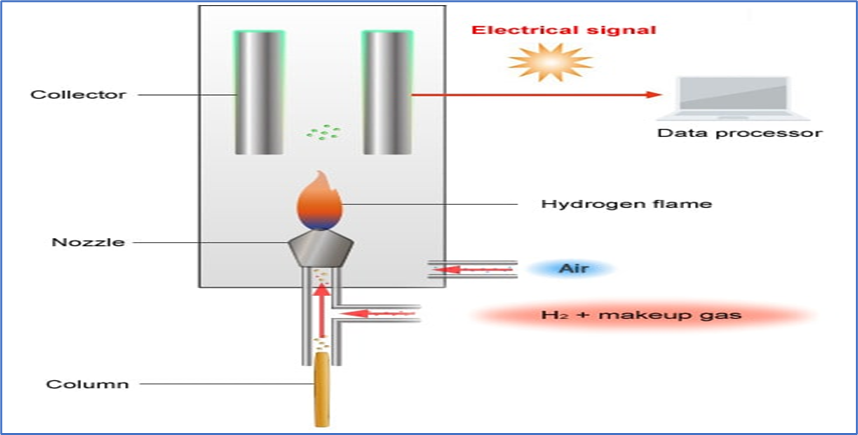
1. Flame Ionization Detectors (FID) :
- In gas chromatography, the FID is the most popular detector.
- The Flame Ionization Detectors (FID) is sensitive to, and capable of detecting, substance that contain carbon atoms (C), which make up nearly all organic substances.
- Carbon atoms having a double bond to oxygen, such as those found in carbonyl and carboxyl groups (CO, CO2, HCHO, HCOOH, CS2, CCl4, etc.) are not sensitive to the FID.
- Working Principle:
- Analyst substances are burned in a hydrogen-air flame.
- The hydrogen flame oxidizes the carbon in a sample carried into the FID detector on carrier gas, causing an ionization reaction.
- A collector electrode attracts the generated ions to an electric field. A signal is generated by counting the number of ions that hit the collector.
2. Thermal Conductivity Detector (TCD)
- Mechanism:
- A detector cell contains a heated filament with an applied current.
- As carrier gas-containing solutes pass through the cell, a change in the filament current occurs.
- A heated filament with an applied current is contained within a detection cell. The filament current changes as a carrier gas containing solutes travels through the cell.
- The current change is compared to a reference cell’s current. A signal is generated after the difference is measured.

3. Electron Capture Detectors (ECD)
- For electrophilic substances, the Electron Capture Detector (ECD) is a selective, high-sensitivity detector. The ECD can detect organic halogen substances, organic metal compounds, diketone compounds, and other organic molecules.
- Mechanism:
- A 63Ni foil lining the detector cell provides electrons. In the cell, a current is generated.
- The current is reduced when electronegative substances collect electrons. A signal is generated once the quantity of current loss is indirectly assessed.

4. Flame Thermionic Detector (FTD)
- It is ideal for organic nitrogen compounds and inorganic and organic phosphorus compounds
- Mechanism:
- The FTD reads the change in ion current obtained at the collector to detect ions.
- When a current is supplied through a platinum coil with an alkali source (rubidium salt) attached, the coil heats up, causing plasma to form around the alkali source.
- A current flows when ions collected in the collector.

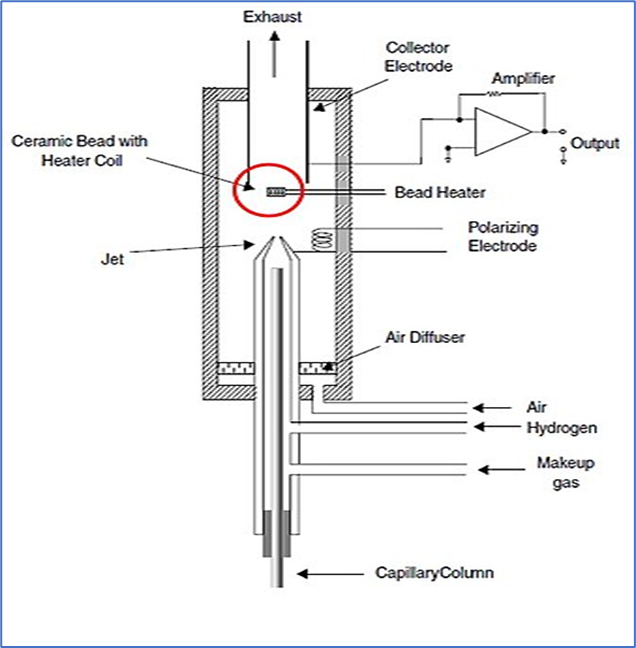
5. Nitrogen-Phosphorus Detector (NPD)
- Also know as Thermionic specific detector (TSD)
- Mechanism:
- Compounds are burnt in a plasma that surrounds a rubidium or cesium bead, that receives hydrogen and oxygen.
- Compounds containing nitrogen and phosphorus produce ions that attract the collector.
- A signal is generated by counting the number of ions that hit the collector.
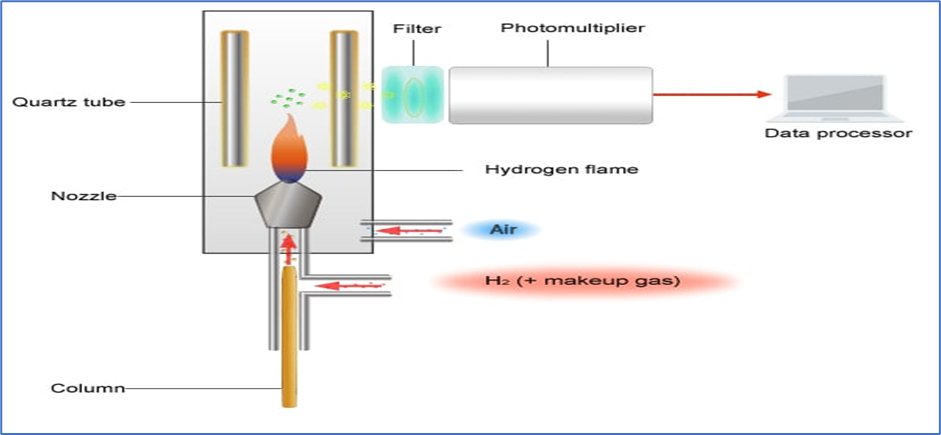
6. Flame Photometric Detector (FPD)
- It is capable of detecting phosphorus (P) compounds, sulfur (S) compounds, and organic tin (Sn) compounds. The Flamephotometric (FPD) is highly selective as it detects element-specific light emitted within a hydrogen flame.
- Mechanism:
- A H2-O2 flame is used to burned Compounds. Sulfur (S) and phosphorous (P) containing compounds produce light-emitting species (sulfur at 394 nm and phosphorous at 526 nm).
- A monochromatic filter allows only one of the wavelengths to pass. A photomultiplier tube is used to measure the amount of light and a signal is generated.
- A different filter is required for each detection mode.
- The photomultiplier tube then converts the detected light intensity into an electrical signal.
7. Photo-Ionization Detector (PID)
- Mechanism:
- High-energy photons emitted from a lamp bombard compounds eluting into a cell.
- Ionization occurs in compounds with ionisation potentials lower than the photon energy. The ions are then attracted to an electrode, measured, and a signal is produced.

8. Electrolytic conductivity detector (ELCD)
- Mechanism:
- Compounds are combined with a reaction gas and pushed through a reaction tube at a high temperature. The reaction products are mixed with a solvent and passed through an electrolytic conductivity cell.
- A signal is obtained by measuring the change in the solvent’s electrolytic conductivity.
- The temperature of the reaction tube and the solvent used decide which chemicals are identified.

9. Barrier Discharge Ionization Detector (BID)
- It can detect all inorganic and organic compounds except, He and Ne.
- Principle of detection:
- The Barrier Discharge Ionization Detector generates a stable Helium (He) plasma, uses the energy produced by the excited Helium (He) to ionize substances, then attracts these ions to a detector’s collector.
- The plasma energy emitted by Helium (He) is incredibly high, capable of ionizing all compounds except He, which is used to form the plasma, and Ne, which has a very high ionization energy.
- As a result, it can detect any substances, other than He and Ne.

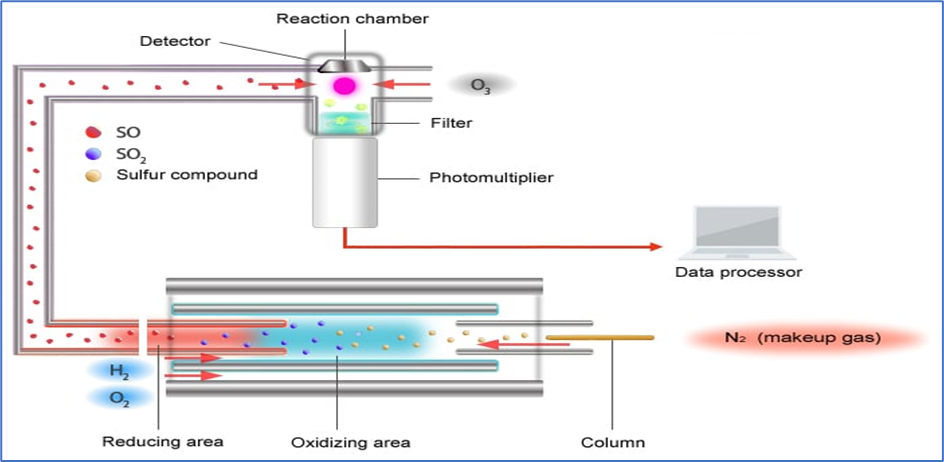
10. Sulfur Chemiluminescence Detector (SCD)
- Sulfur chemiluminescence Detector (SCD) is one of Gas Chromatography Detector used for sulfur contain substance mainly.
- Mechanism:
- The chemiluminescence reaction generated by ozone oxidation is employed by the sulphur chemiluminescence detector (SCD).
- Inside an extremely high temperature (about 1000 °C) oxidative-reductive furnace, sulphur compounds are transformed to an X-S chemical species (mostly SO) capable of chemiluminescence.
- The chemical species X-S is delivered to the detector area, where it is converted to an excited-state SO2* by ozone (radical). When SO2* returns to its base state, it emits light, which the SCD detects by measuring this light with a photomultiplier tube.
11. Gas chromatography–Mass Spectrometry (GC-MS)
- GC-MS is more advanced Gas Chromatography Detector for identified the impurity.
- Mechanism:
- The detector is kept under vacuum. Analysts (Sample) are bombarded with electrons (EI) or gas molecules (CI). Charged ions or fragments are formed when compounds fragment.
- These ions are focused and accelerated into a mass filter. The mass filter selectively permits all ions of a precise mass to pass through to the electron multiplier.
- All ions with a particular mass are identified. The mass filter then lets the next mass through while rejecting all others.
- Several times per second, the mass filter scans progressively through the defined range of masses.
- For each scan, the total number of ions is counted. The chromatogram is created by plotting the number of ions per scan against time. For each scan, a mass spectrum is generated, which shows the various ion masses against their abundance or number.
- Selectivity refers to a compound’s ability to produce fragments within a given mass range. May be an inclusive range of masses (full scan) or only select ions (SIM)

Read More:
