Naming System for Biosimilar Products
Biosimilars are biological medicinal products that are highly similar to an already approved
reference biologic. Due to the complexity of biological products, biosimilars are not identical
copies, which makes clear and region-specific naming systems essential for
traceability, pharmacovigilance, and patient safety.
Why Biosimilar Naming Matters
- Ensures accurate pharmacovigilance
- Prevents medication errors
- Allows effective batch traceability
- Supports regulatory oversight
Global Concept of INN
The International Nonproprietary Name (INN), assigned by WHO, identifies the active
substance. For biosimilars, the core INN remains the same as the reference product,
while regional regulators apply additional naming rules.
Reference Biologic
Biosimilar 1
Biosimilar 2
Same INN – Regional Naming Differences
US FDA Naming System
The US FDA assigns a distinct nonproprietary name consisting of:
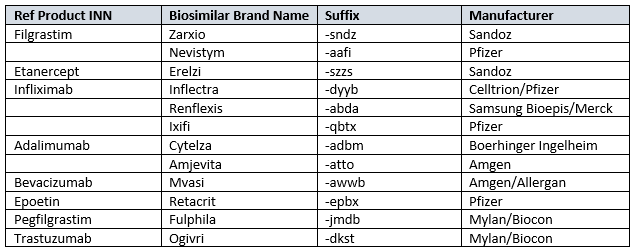
FDA-Approved Real Examples
| Product Type | Nonproprietary Name | Brand Name |
|---|---|---|
| Reference | Filgrastim | Neupogen |
| Biosimilar | Filgrastim-sndz | Zarxio |
| Biosimilar | Adalimumab-atto | Amjevita |
| Biosimilar | Trastuzumab-dkst | Ogivri |
Adalimumab
+
– atto (FDA suffix)
Some of Examples:
European Union (EMA) Naming System
The European Medicines Agency (EMA) uses the same INN for reference biologics and
biosimilars. No suffix or qualifier is added to the nonproprietary name.
| INN | Brand Name | Company |
|---|---|---|
| Infliximab | Remicade | Janssen |
| Infliximab | Remsima | Celltrion |
| Infliximab | Inflectra | Pfizer |
Indian (CDSCO) Naming Practice
India follows a same INN approach for biosimilars and reference biologics.
Differentiation is achieved through brand name, manufacturer, and batch number.
| INN | Brand Name | Manufacturer |
|---|---|---|
| Adalimumab | Exemptia | Zydus |
| Trastuzumab | Canmab | Biocon |
| Bevacizumab | Bevatas | Intas |
Regional Comparison Summary
| Region | INN Usage | Suffix / Identifier |
|---|---|---|
| US FDA | Same INN | Mandatory 4-letter suffix |
| EMA | Same INN | None |
| India (CDSCO) | Same INN | None |
| Japan | Same INN | “BS” notation |
Regulatory Insight
It is a regulatory mechanism to ensure traceability, safety monitoring, and clear product identification.
Read More:
